
Chapter 14 The Ideal Gas Law and Kinetic Theory. To facilitate comparison of the mass of one atom with another, a mass scale know as the atomic mass scale. - ppt download

Monoatomic Gas: Made of one atom, like HeMonoatomic Gas: Made of one atom, like He Diatomic Gas: Made of two atoms, like Cl 2 or H 2Diatomic Gas: Made. - ppt download

One mole of an ideal gas at standard temperature and pressure occupies 22.4 L (molar volume). What is the ratio of molar volume to the atomic volume of a mole of hydrogen ? (
The chemistry of agriculture, for students and farmers. Agricultural chemistry. 314 THE CHEMISTRY OF ANIMAL PHYSIOLOGY
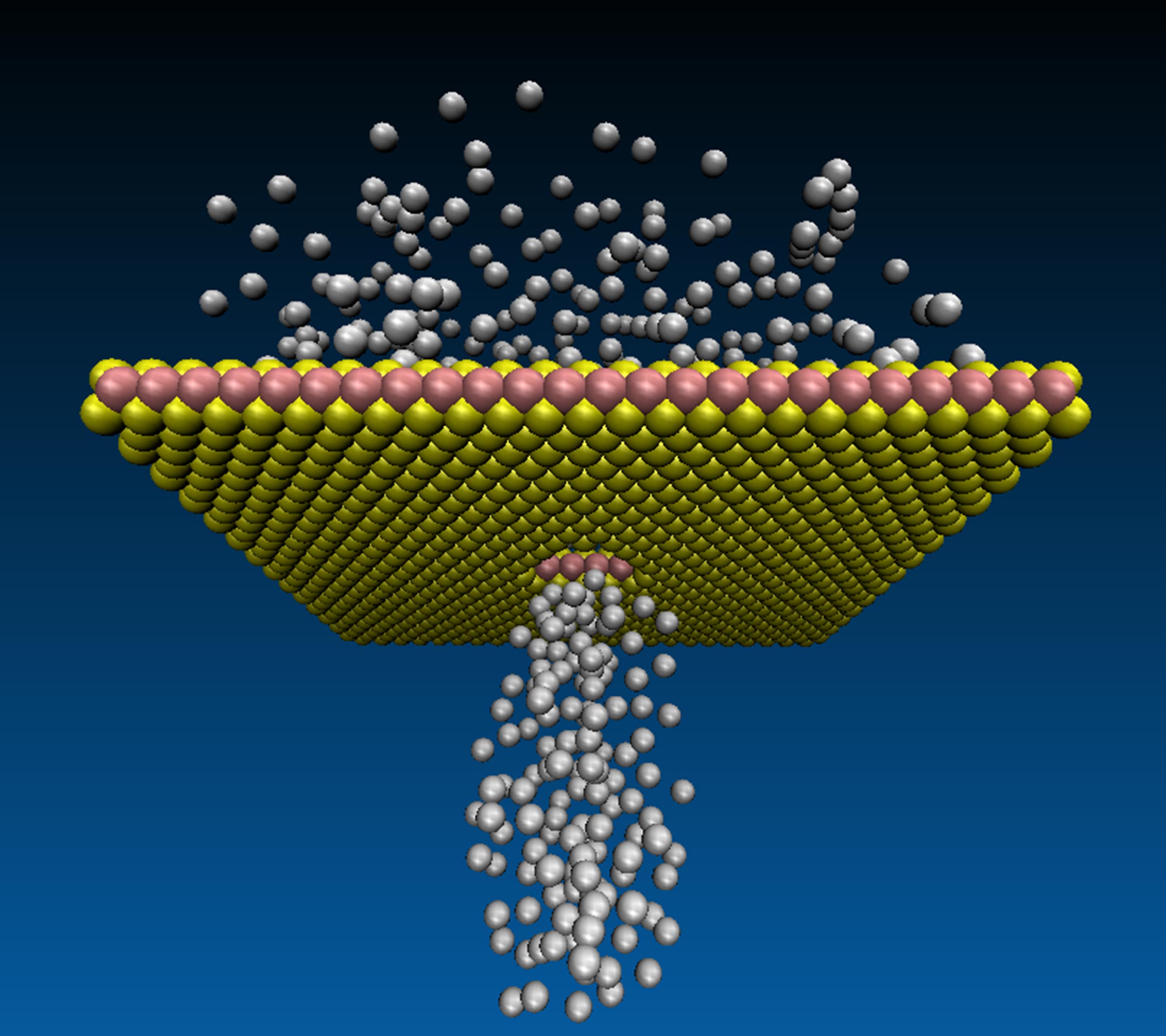
Ultra-Fast Gas Flows Through Tiniest Holes in One-Atom-Thin Membranes – Validates Century-Old Equation of Fluid Dynamics
The weight of 350ml of diatomic gas at 0°C and 2 atm pressure is 1 g. The weight of one atom is? - Quora

Chapter 14 The Ideal Gas Law and Kinetic Theory of Gases Slideshow and powerpoint viewer: 14.1 Molecular Mass, the Mole, and Avogadro's Number • The atomic mass scale facilitates comparison of

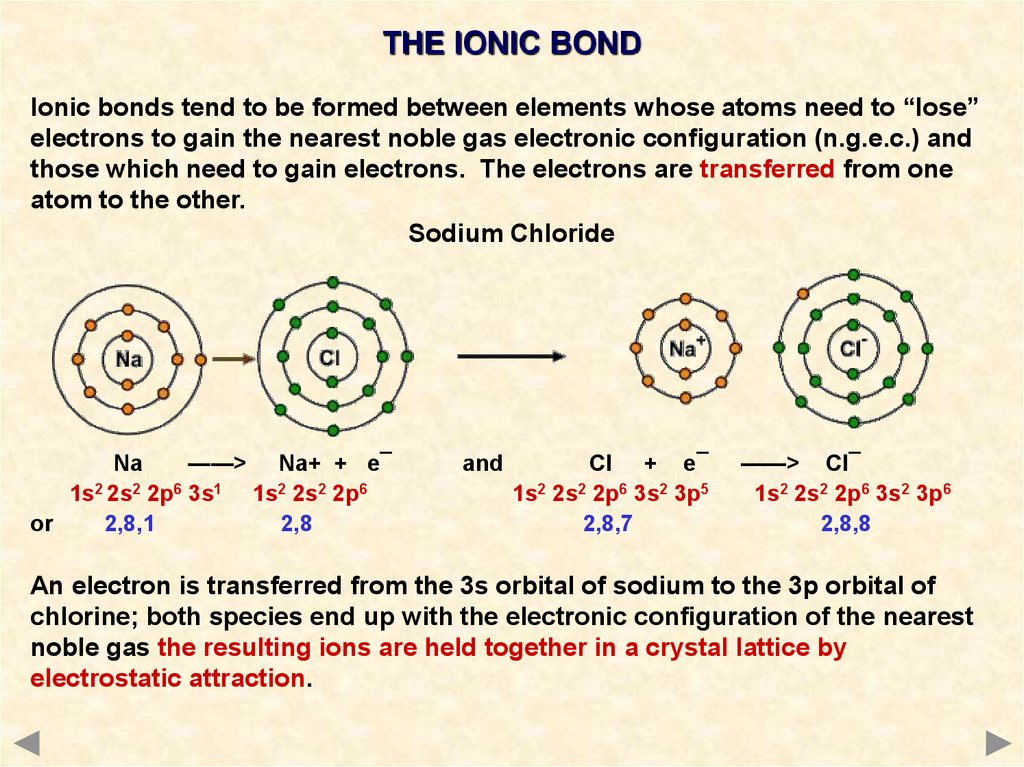


/186450350-56a132cb5f9b58b7d0bcf751.jpg)










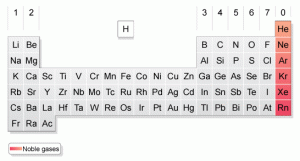
/GettyImages-545864473-5a769a63119fa8003740347a.jpg)

