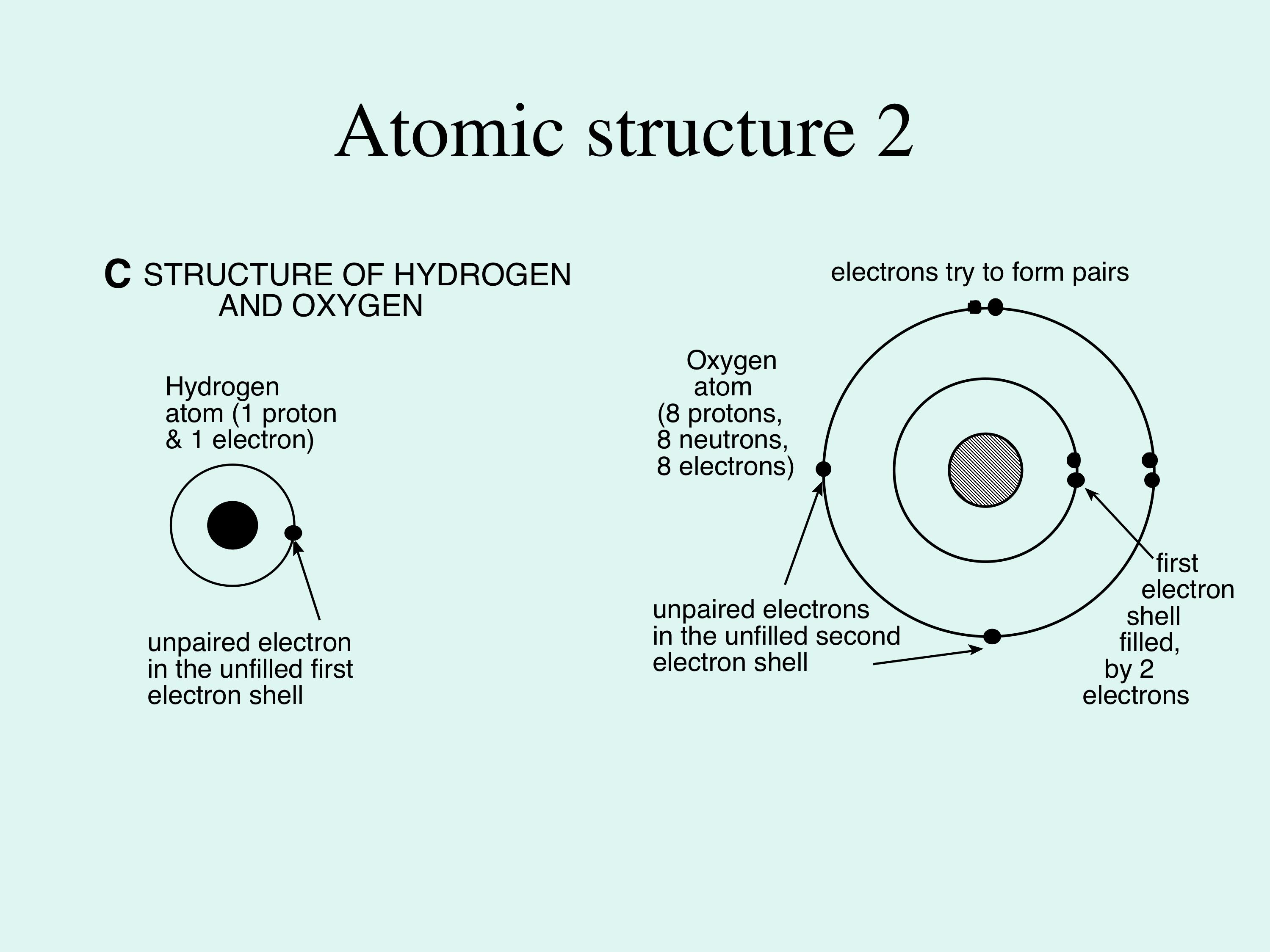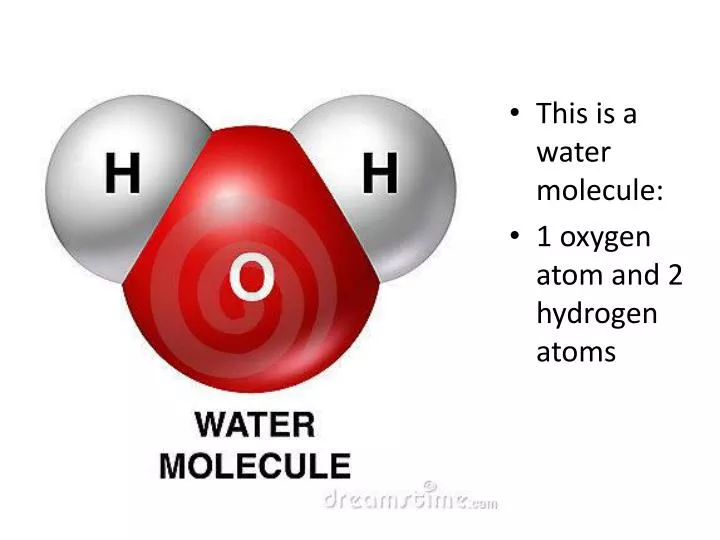
PPT - This is a water molecule: 1 oxygen atom and 2 hydrogen atoms PowerPoint Presentation - ID:2595469
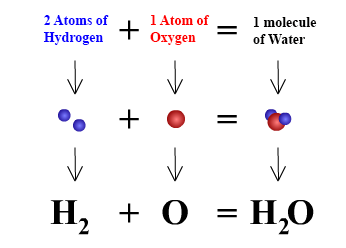
Water is made of 2 atoms of hydrogen and one atom of oxygen then why doesn't water catch fire because both the above materials are highly combustible? - Quora

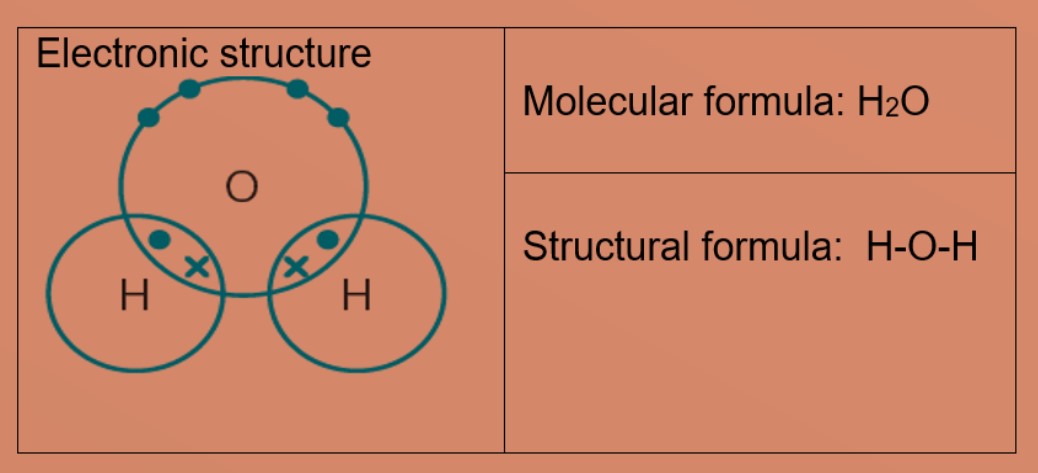
One molecule of water contains 2 atoms of hydrogen and 1 atom of oxygen The atomic masses of hydrogen - Maths - Linear Equations in Two Variables - 10034561 | Meritnation.com

Why does combining hydrogen and oxygen typically produce water rather than hydrogen peroxide? - Scientific American

1 molecule of water contains 2 atoms of Hydrogen and 1 atom of oxygen. Hence the Volume of hydrogen is - Brainly.in